Life Innovation Achieved
through Co-creation
Of the estimated 6,000 intractable or rare diseases that have been identified, it is said that 80% are genetic in origin. One such disease is amyotrophic lateral sclerosis (ALS), which in some cases is inherited, and is known for having claimed the life of legendary baseball player Lou Gehrig at the young age of 37. ALS is one of thousands of intractable diseases that sentence victims to a shortened future, during which physical and mental capabilities gradually decline. For decades, there had been few sources of hope for patients with rare diseases. With recent technological and scientific developments, however, it appears there may be cause for optimism.
Returning from the Point of No Return
― Technology enabling the restoration of cells to an embryonic state represents an historic achievement in the quest to cure rare diseases
Thirty-seven was the age at which Lou Gehrig died; it also marks Michelangelo’s age when he put the finishing touches on the ceiling at the Sistine Chapel, and Charles Dickens’ age when David Copperfield was published. In degrees centigrade, it represents the average normal body temperature. Expressed in trillions, it is the estimated number of cells that comprise the human body.
It all begins with a single fertilized egg, or zygote, which divides and then multiplies, the resulting cells clumping together to form the embryo. The embryonic stem (ES) cells that constitute the early embryo differentiate into specialized cells such as skin, bone, blood, and heart. Once differentiated, they cannot return to their former state or be repurposed. Cardiac muscle cells and matured neurons are also incapable of regeneration.
In 2006, a team led by Shinya Yamanaka, then a professor and researcher at Kyoto University’s Institute for Frontier Medical Services, succeeded in transforming skin cells of laboratory mice into cells extremely similar to ES cells through the introduction of four transcription factors (narrowed down from more than one hundred possibilities and exponentially more permutations). These are proteins that, in effect, tricked the cell into believing it needed to adapt to an embryonic environment. A little more than a year later, they achieved the same result using human skin fibroblasts in a discovery that scientists had previously thought unachievable.
These cells were named induced pluripotent stem (iPS) cells, and are virtually indistinguishable from ES cells. Both are pluripotent, meaning not only can they regenerate themselves; they can be differentiated into specialized cell types through treatment with growth hormones and other agents. iPS cells can also be reproduced with no volume restrictions. This achievement created tremendous expectations for application in regenerative medicine and drug development.
“The history of iPS cell research has only just begun, and this technology has a
remarkable potential for use in cell therapy, drug screening and personalized
medicine.”
Shinya Yamanaka (Director, Center for iPS Cell Research and Application, Kyoto
University)
Nobel Lecture, December 7, 2012

While the enactment in Japan of the Act on Safety of Regenerative Medicine (also known as the RM Act)—after nearly two decades of work and discussion—represented a monumental step forward in the development of a regenerative medicine infrastructure, many obstacles still need to be negotiated before iPS cells can contribute significantly to regenerative medicine. Paradoxically, the RM Act contributed to the stoppage of projects that had been in progress, such as cell transplantation for patients suffering from age-related macular degeneration (deterioration of eyesight, until now considered irreversible). The team undertaking this project essentially lost an entire year. This project and others are further weighed down by prohibitive costs—in the case of the macular degeneration transplant project, one attempt incurred costs on the order of US$1 million.
The potential of iPS cells with regard to the development of new drugs, however, is presently very promising. Tests to confirm the efficacy, toxicity, and safety of new drug candidates on human cells—as well as in the development of new drugs to be used in the treatment of heretofore incurable and irreversible intractable diseases—can be conducted using iPS cells. This has made it possible to circumvent ethical concerns surrounding the use of human embryos in the derivation of ES cells, and the use of animals in clinical testing. It also has the potential to greatly accelerate the process. Developing a drug can take up to ten years and incur costs in the billions of yen, and until now new drug candidates have not been tested on human cells until virtually the end of the process. iPS cells make it possible to perform testing on human cells at a much earlier stage of development, thereby weeding out candidates that are not compatible with human cells before having invested considerable time and funds.
Progress has been achieved in dealing with the treatment of particular diseases, and as such a palpable shift in focus is underway toward rare diseases which affect a relatively small number of people, and to high unmet medical needs including cancer and diseases related to the central nervous system. Increasing momentum in next-generation R&D based on new technological innovation targeting disease fields where onset and progression mechanisms remain unknown will lead to the aggressive application of a host of new technologies.
Leading Industry and Society into a New Age
― Yokogawa’s superior technological capabilities and passion for collaboration position it as a leader in the era of life innovation
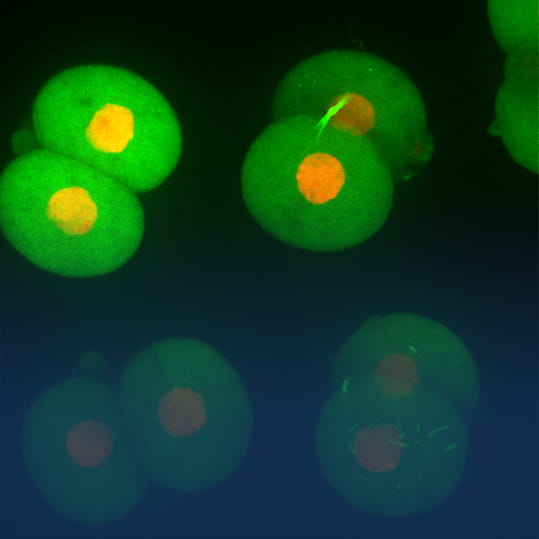
Yokogawa has integrated its core cell imaging technology with the measurement, control, and information technologies that have long been its strengths, to realize cell-based new drug screening technology. The company’s involvement in biotechnology research goes back to the late 1980s when then-chairman Shouzo Yokogawa issued a mandate to the R&D department to apply its considerable expertise to the measurement of a “soft” target. Perhaps motivated in part by the discovery in 1961 of green fluorescent protein, or GFP—which, decades later, would earn Dr. Osamu Shimomura the Nobel Prize in Chemistry—the researchers chose to investigate cells.
Through communication between the research staff and colleagues at universities and external research institutions, Yokogawa determined that the need existed for high-definition, high-sensitivity imaging of live cells. This proved to be the impetus for the R&D activity that took place over the next decade, with the objective of developing a device that would enable spatiotemporal live cell imaging.
In 1996, Yokogawa released the first incarnation of its series of confocal scanner devices, the CSU10. It enabled the high-speed acquisition of confocal images using raster scanning which disperses hundreds to thousands of low-power laser beams, successfully reducing the damage done to living cells under examination and facilitated extended observation. The company didn’t stop there, however. Over the ensuing twenty-plus years, discussion, research, and collaboration produced increasingly evolved versions of the CSU. Today, the CSU series is the de facto industry standard, having sold about 3,000 units worldwide.
In the early 2000s, CSU series confocal scanner sales were concentrated in biological and medical research. Yokogawa began to look at other fields where the technology could be applied, and eventually decided on the high-content analysis (HCA) market. HCA, a method for assessing the status of a cell based on information such as size, shape, and protein expression acquired from the cell itself, had been developed in the late 1990s. This would bring together the capability to acquire confocal images provided by the CSU, and technology to make possible the analysis of those images.
“Using human cells at a very early stage of drug development will help to identify
compounds which do not work in human cells.”
Oliver Brüstle (Head, Institute of Reconstructive Neurobiology, University of Bonn
Medical Center)
Video: “Stem cells - the Future: an introduction to iPS cells”

These efforts resulted in the 2009 release of the CellVoyager CV6000, a high-throughput cytological discovery system that employed the HCA method. In conjunction with a CSU, the CV6000 enabled simultaneous observation of multiple samples at industry-leading speed and resolution to detect changes in the number of target molecules, cellular kinetics and morphology, and more. The trend toward automating and accelerating the new drug development process—during which candidate compounds requiring screening can number in the millions—is picking up steam. Yokogawa’s technology provides valuable support to these efforts in the form of reduced manhours and cost, and superior resolution for clear viewing which facilitates accurate efficacy confirmation and helps to ensure safety. The accelerated speed also potentially contributes to more rapid treatment of patients suffering from that particular disease.
In 2010, one of the world’s preeminent medical research institutions, the German Center for Neurodegenerative Diseases (DZNE) selected Yokogawa’s CV6000 as the key element of its drug discovery support system development project, and DZNE and Yokogawa also worked together to further advance the technology. Over the next several years, the company released increasingly evolved models in the series. These new models responded to demands for a broader imaging area and higher throughput. They also offered benefits such as the ability to observe and analyze individual cells and cell clusters; to measure cell characteristics and compile data toward discovering the optimal environment for cultivation; and to confirm the quality and extent of differentiation of cells.
Yokogawa’s devices and systems have come to fruition through close collaboration with users, and were designed to aid them in achieving their goals from the perspectives of the users. This is the philosophy that has driven Yokogawa’s tireless efforts to improve quality, speed, and ease of use.
Yokogawa is pursuing development in single-cell mass spectrometry and single-cell gene analysis to achieve precise analysis of individual cells, which is not possible through imaging. The company established the Single Cellome Co-innovators Consortium in conjunction with the University of Shizuoka in April 2018. Based at the Shonan Health Innovation Park in Kanagawa Prefecture southwest of Tokyo, the consortium’s objective is to apply the combined accumulated expertise in single-cell mass spectrometry to the development of advanced systems that will enable detailed examination of individual cell interiors. The systems are expected to help accelerate new drug development.
Yokogawa is lending its support to efforts related to the application of iPS cells to drug development. It is among several private-sector entities collaborating on research connected to a special program as part of The University of Tokyo’s Future Society Initiative. The primary aim of the program is the formulation of a system to evaluate drug efficacy and toxicity through academic-industrial collaboration, mainly through the use of various human cells derived from human iPS cells.
The sophisticated technologies Yokogawa has developed in the three-plus decades since it began its foray into life science have the power to make major contributions to maximizing the value and realizing the potential of iPS and ES cells in new drug development and, in the future, regenerative medicine. The successful development of new drugs will require global-scale collaboration in the sharing of intelligence and data. Yokogawa’s co-innovative mindset enables the company to work successfully and collaboratively with the most advanced partners, resulting in synergy that will lead to unprecedented achievements in the life sciences field. Armed with some of the most advanced technological capabilities in the field, Yokogawa will contribute to the development of the industries that provide the foundation for healthy, comfortable lives for people around the world.