3D spheroid/organoid models are becoming the new standard for drug discovery applications because of their physiologically relevant characteristics relative to 2D cultured cells. However, in order that the spheroids/organoids function properly, it is necessary for the cells and the extracellular matrix (ECM) to be organized in the appropriate spatial arrangement to form microstructures, and how to create 3D spheroid models which mimic the environment in a human body is a hot topic. Nobuhiko Kojima and his group at Yokohama City University, Japan, are developing the technologies of designing different types of spheroids with internal microstructures.

Introduction of our research
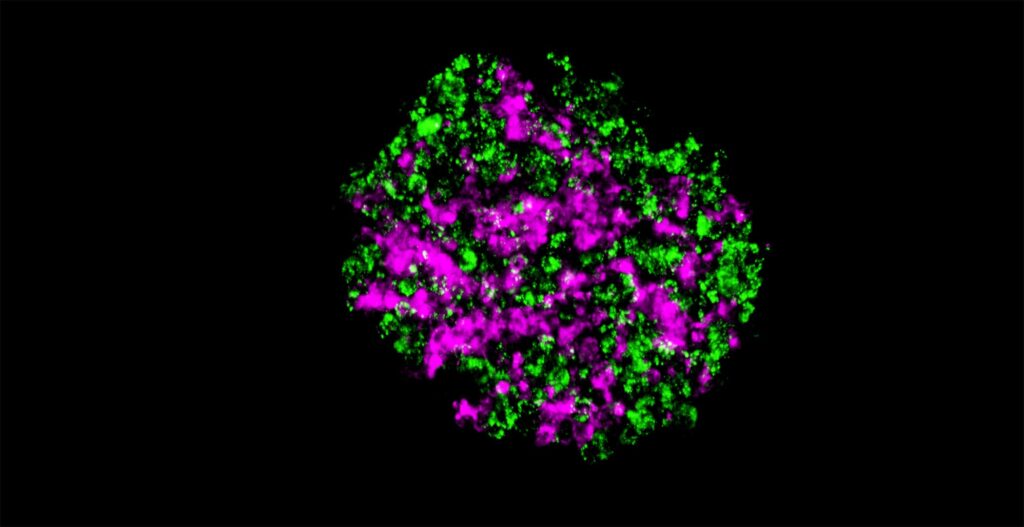
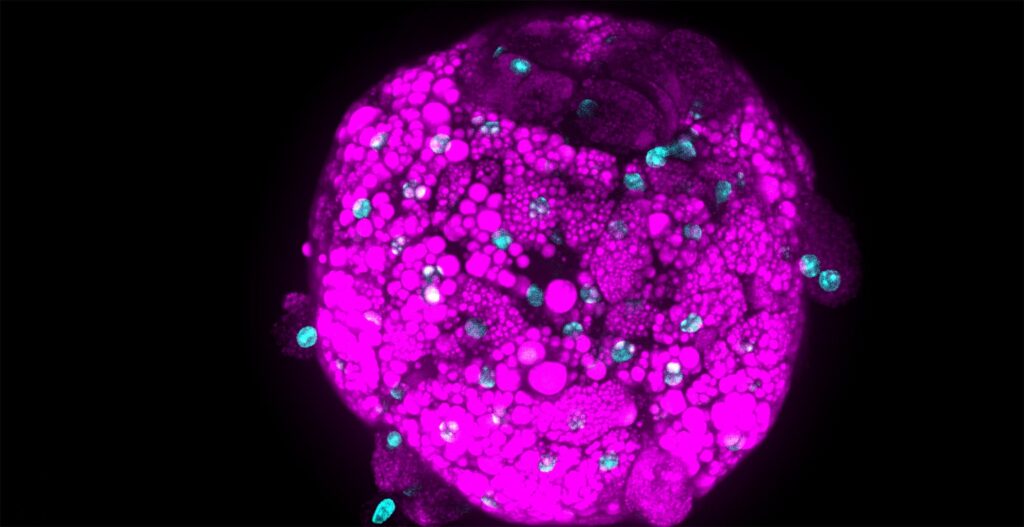
We are studying how to make spheroids that have various cell aligning patterns (Figure 1). Using the forced cell aggregation method that utilizes the swelling of methylcellulose containing medium, suspended cells can be aggregated in about 10 minutes [1]. Forcibly aggregated cells can slowly but certainly form cell-cell adhesions, so our methods realize fabricating spheroids consisted of cells with very low adhesive properties. For example, we were able to form spheroids using suspended bone marrow cells [2]. In addition, it is possible to aggregate particulate materials with a diameter from about 100 nm to at least 100 µm in about 10 minutes [1]. It means that we can make hybrid spheroids comprised of hydrogel beads and cells. If we replace 50% of cells in the spheroids with hydrogel beads, the inside of the spheroids shows channel-like structures [3]. Furthermore, since extracellular matrix (ECM) can be aggregated with the cells, it is possible to create spheroids in which a thin-layered ECM is filled inside [4]. Spheroids filled with ECM enable the production of more physiological spheroids. For example, in type 1 diabetes, it is known that hyaluronic acid accumulates in the islets. Our methods can create a type 1 diabetes model islet by aggregating both islet cells dissociated from healthy islets and hyaluronic acid. Examination of this model islet revealed several novel insights in the direct effects of hyaluronic acid on the islet cells.

CellVoyager CQ1 is used to evaluate the spheroid quality
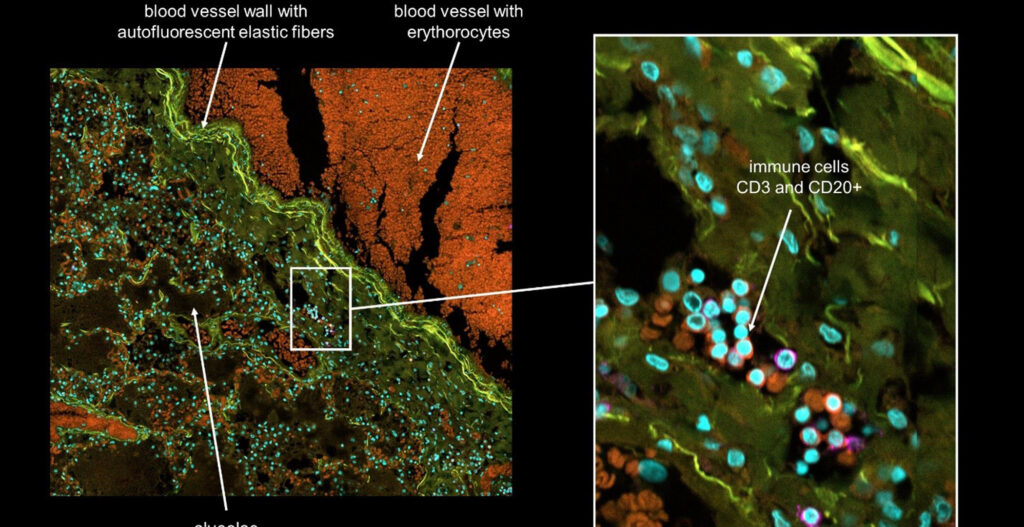
To create a unique spheroid production method, it is necessary not only to propose a production process but also to objectively evaluate the produced spheroids. We are conducting confocal laser observation with the CellVoyager CQ1 to confirm the internal structures of the produced spheroids. For example, we measured the thickness of ECM filled into the spheroids in a recent work [4]. The CQ1 is also used to evaluate cytotoxicity by detecting living and dead cells after exposing the spheroids to drugs.
We monitor how microstructures are formed in a living spheroid with the CQ1
Since a CQ1 is equipped with a CO2 incubator system, it is possible to observe the structures inside of spheroids in a time-lapse manner by using fluorescently labeled cells and ECMs. This is indispensable in our laboratory because spheroids consisting of multiple types of cells and ECMs often alter their structures in a self-organizing manner (Figure 2). The CQ1 can automatically observe their morphological changes at appropriate intervals even at midnight. It is also excellent that the observed results can be analyzed quantitatively. And most of all, Yokogawa has experienced staff who can support and help us quickly when problems arise.
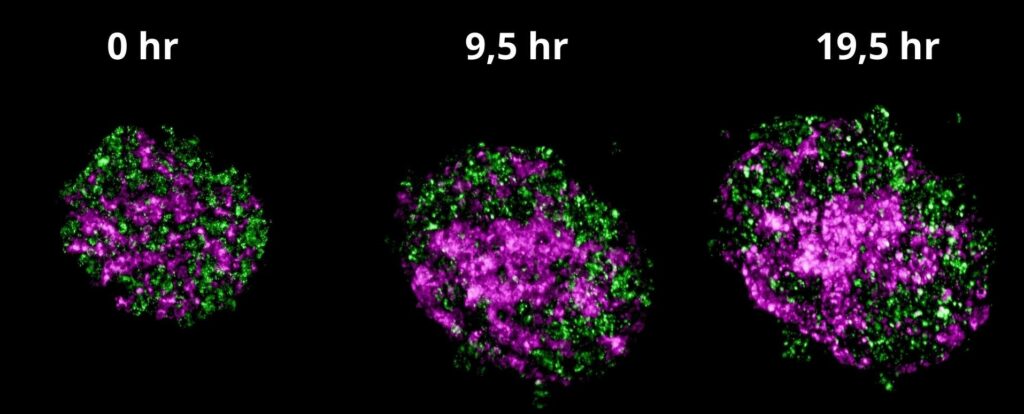
Figure 2 a) Time-lapse movie of spheroid self-organization. 1.5 hours after cell suspension was ejected into methylcellulose containing medium, image acquisition was performed on a CQ1 for 19 hours at 0.5-hour intervals.
Figure 2. b) Representative spheroid images (max intensity projection) at 0, 9.5, and 19.5 hours of the time-lapse assay. Two different types of cells, HepG2 cells (green) and HUVECs (magenta) which were initially random, are observed to self-organize with the network.
Outlook on techniques of designing 3D spheroid/organoid models for drug discovery
3D models such as spheroids are becoming indispensable for drug discovery and regenerative medicine. We expect that our methods, which enable us to produce spheroids with various designs, can be critical techniques to accomplish many purposes. We would like to improve the spheroid production methods with the aim of realizing a society where everyone can live in good health.
References
[1] Kojima, N., Takeuchi, S. and Sakai, Y. Rapid aggregation of heterogeneous cells and multiple-sized microspheres in methylcellulose medium. Biomaterials, 33, 4508-4514 (2012).
[2] Sayo, K., Aoki, S. and Kojima, N. Fabrication of bone marrow-like tissue in vitro from dispersed-state bone marrow cells. Regen. Ther., 3, 32-37 (2016).
[3] Kojima, N., Takeuchi, S. and Sakai, Y. Fabrication of microchannel networks in multicellular spheroids. Sensor. Actuat. B-Chem., 198, 249-254 (2014).
[4] Tao, F., Sayo, K., Sugimoto, K., Aoki, S., and Kojima, N. Development of a tunable method to generate various three-dimensional microstructures by replenishing macromolecules such as extracellular matrix components and polysaccharides. Sci. Rep. 10, 6567 (12 pages) (2020).