Original publication from PharmaManufacturing.com.
Innovative Technology for Active Pharmaceutical Ingredients(APIs)
1 Changes in manufacturing technology for active pharmaceutical ingredients
In the active pharmaceutical ingredients (APIs) field, batch manufacturing is controlled for each process, and continuous manufacturing has not been incorporated. Since stable supply must also meet strict quality regulations, research has been conducted on the technological shift from batch manufacturing to continuous manufacturing, but the practical application has not been actively undertaken.
However, in recent years, as the impact of human error on production activities has become apparent and it has become difficult to secure a labor force, continuous manufacturing is increasingly advantageous since it does not require taking products out of processes, can reduce manufacturing time and labor, and can be expected to reduce human error. Since the production volume is adjusted according to the operating time of the equipment, it is possible to flexibly respond to demand fluctuations. In addition, since there is no need for procedures to scale up while changing conditions in different facilities as in the past, it also leads to a shortening of the development period, which makes a smooth transition from investigational drug production to commercial production possible. Another advantage is that real-time monitoring of quality during the process enables highly reliable quality assurance.
Continuous manufacturing has already been introduced in fields such as petrochemicals and food, but progress has been slow in the pharmaceutical industry due to regulatory constraints. However, due to its merits, the guidance released by the U.S. Food and Drug Administration (FDA) in 2004 included the application of continuous manufacturing to pharmaceuticals, and in 2015 the use of drugs manufactured in continuous manufacturing. These were the first solid preparations approved by the FDA. The International Conference on Harmonisation of Pharmaceutical Regulations (ICH) began work on formulating guidelines in 2018 and continues working to improve the environment for practical applications.
2 What is continuous manufacturing?
Conventional batch manufacturing is a production method in which raw materials are input into manufacturing equipment and the products are taken out immediately after the completion of the single-process manufacturing operation. In this method, the operation stops for every single process, and the operation of raw material input, manufacturing (reaction/purification), and product discharge is repeated. Consequently, the operation is complicated and requires manpower.
Continuous manufacturing, on the other hand, is a method in which raw materials are continuously injected into the manufacturing facility and the product is continuously taken out while the manufacturing process is in operation. This streamlines the entire production facility’s operation and reduces the human burden by automatically controlling and optimizing multiple processes (see Figure 1).
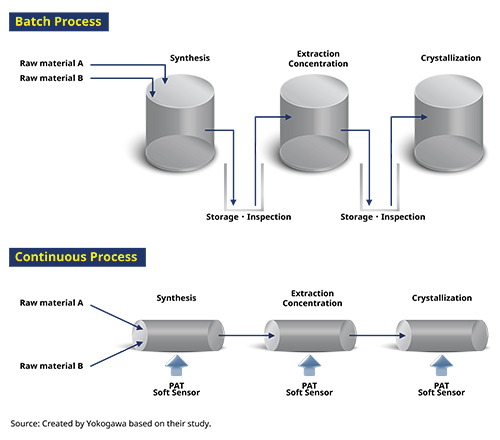
Figure 1: Batch versus Continuous Process
In continuous manufacturing of APIs, operations such as reaction and crystallization are connected and automated, and these processes are controlled to save labor and improve productivity. The time required to scale up and down the production of active pharmaceutical ingredients and adjust parameters and operations for each production scale shortens, making the early market introduction of active pharmaceutical ingredients and final products possible.
The growing demand for formulation quality is also increasing the demand for the quality of APIs. Fortunately, continuous manufacturing’s constant monitoring of the manufacturing process also minimizes the risk of missing parts by improving and stabilizing quality. In addition, reducing the amount of solvent used in the manufacturing process is necessary, minimizing environmental impact by achieving a high reaction rate, and reducing workplace risks by improving manufacturing facilities. Efforts to realize these opportunities are becoming possible via the practical application of continuous manufacturing technology.
3 Technologies necessary to realize this
To achieve continuous manufacturing, it is necessary to fully understand the product and the manufacturing process to ensure a stable state.
Since the reaction mechanisms differ between batch manufacturing and continuous manufacturing, it is imperative to develop a catalyst suitable for continuous manufacturing and a process system engineering management strategy to maximize production efficiency. Furthermore, since continuous manufacturing does not stop production, measurement technology that monitors the manufacturing process in real time is essential. In particular, Process Analytical Technology (PAT) by Raman and Near Infrared Spectroscopy (NIR) create Real Time Release Testing (RTRT). In addition to PAT, information that is difficult to measure is estimated in real-time using simulation technology. Soft sensor technology attracts attention. In the manufacturing process, while variables such as temperature, pressure, and flow rate can be measured with sensors, concentration and density must be analyzed as samples. The time-consuming analysis prevents advanced control. Soft sensor technology can build algorithms based on past data and can estimate changes in response. It is also important to have an information server that integrates, controls, monitors, and manages information throughout the manufacturing process. In the continuous manufacturing process, a Programmable Logical Controller (PLC) controls each step such as reaction, extraction, and crystallization. Consequently, it is essential to integrate, monitor, and control information at the top of the entire process.
Furthermore, it is important to integrate and manage not only process information but also quality data to realize highly accurate soft sensors and simulations. In addition, when manufacturing multiple items, managing recipe information is also a top priority.
Digital Transformation (DX) that integrates this scattered information and digitizes factories is essential. Consequently, building systems that can flexibly connect each PLC and higherlevel systems is of utmost importance. NAMUR has taken the lead in developing the Modular Type Package (MTP) and is expected to utilize the standardization framework. The integrated information server smoothly and efficiently executes an optimally integrated operation, monitoring and managing each device from individual optimal operation to total optimization. By utilizing dynamic emulation technology, it is possible to quickly create continuous production of each item, monitor quality, and enhance manufacturing management. By integrating these technologies, continuous production will revolutionize high-mix, low-volume production manufacturing processes and create new value in the manufacturing industry.
4 Development trends
Continuous manufacturing technology is under development globally. In Germany, to create highly flexible module production, the development of MTP is being promoted mainly by NAMUR with a plug-and-produce goal that will affect interoperability by modularizing and connecting multiple manufacturing units. The European Union (EU), the United States, Australia, Japan, and other countries are developing catalysts, equipment, and control technologies. The Pharmacy on Demand project in the U.S. aims to continuously automate all processes from raw material input to tablet processing.
In Japan, the Flow Science and Technology Consortium (FlowST), an industry-academia-government collaboration, was launched in 2015, and in 2021, practical use began mainly by eight private companies.
With pharmaceutical companies, there is an active introduction of continuous manufacturing, mainly by European and American companies (see Figure 2). Eli Lilly and Pfizer have quickly obtained FDA approval for continuous manufacturing, and some drugs are already in commercial production. Novartis of Switzerland has established CONTINUUS Pharmaceuticals based on the results of its joint research with the Massachusetts Institute of Technology, which provides equipment design services for continuous manufacturing. SK biotek of Korea has introduced continuous manufacturing in many batch manufacturing by utilizing the technical capabilities of catalyst and process development cultivated in the petrochemical business for the continuous manufacturing of pharmaceuticals.
In Japan, Shionogi Pharma is planning and promoting capital investment to establish a continuous production system for manufacturing processes, investigational drugs, and active pharmaceutical ingredients. It has also established Pharmira Co., Ltd. with seven private companies, including Yokogawa Electric, to develop continuous manufacturing technology for small molecule active pharmaceutical ingredients and utilize the technology for the CDMO business.
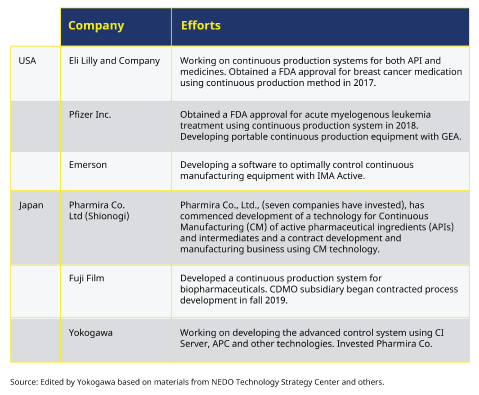
Figure 2: List of companies moving towards continuous process pharma technology
5 Conclusion
There are many technical challenges to fully realizing continuous manufacturing in pharmaceutical manufacturing. However, the merits of continuous manufacturing are enormous, and attention is focused on solving technical problems.
Of course, plant design for continuous manufacturing is important, but control technology to operate it flexibly and efficiently is also very important. Innovative development of control technology will continue to be highly anticipated in the future.
Figure 3: Example of a data-driven optimization solution workflow
Related Industries
-
Pharmaceutical
Under continual pressure to increase profitability, maintain government compliance, and meet emerging market opportunities, the pharmaceutical manufacturing industry faces unique challenges that require unique solutions. As one of the world’s leading industrial automation suppliers, Yokogawa is poised and prepared to deliver those solutions, creating individualized lean manufacturing techniques that deliver consistent, measurable results.
Related Products & Solutions
-
Control and Safety System
Support long-term stable production with flexible solutions
Have Questions?
Contact a Yokogawa Expert to learn how we can help you solve your challenges.