Application: Nano-point Delivery Imaging System
- Tracking Intracellular Delivery and Structure Formation of a Phase-Separating Protein
- Tracking Nuclear Delivery and Phase Separation Disruption by Artificial Antibodies (Monobodies)
- Tracking Antibody Delivery and Intracellular Molecular/Organelle Staining in Live Cells
- Tracking Compound Delivery and Nuclear Translocation
Tracking Intracellular Delivery and Structure Formation of a Phase-Separating Protein
In neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD), dysregulation of liquid–liquid phase separation has been reported. FUS (Fused in Sarcoma) is one of the proteins that undergo phase separation in cells and is thought to be closely associated with these neurodegenerative disorders.
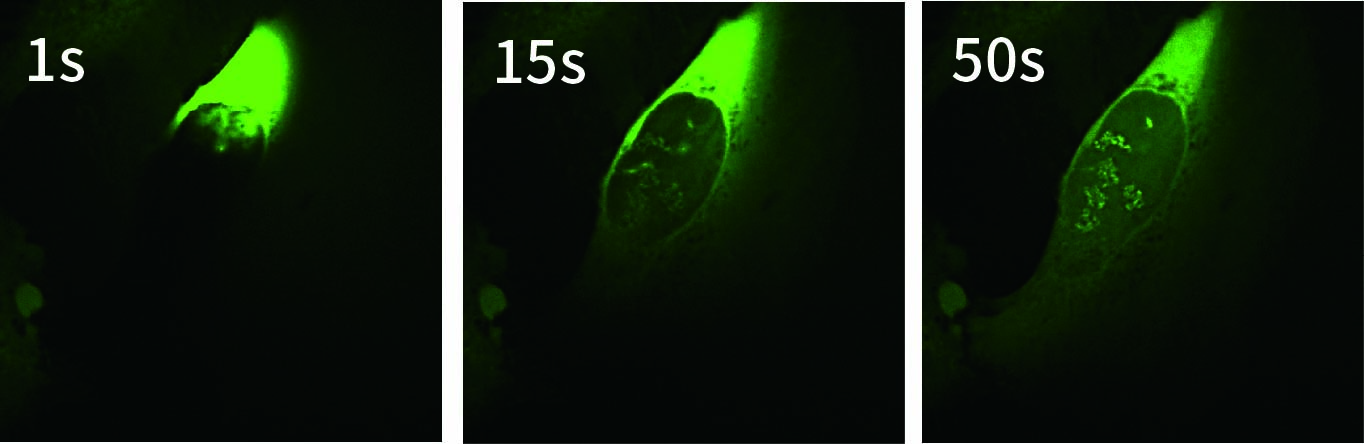
Conventionally, FUS has been analyzed by introducing and expressing its gene in cells. However, this approach has limitations in enabling real-time analysis of the behavior of newly translated FUS protein immediately after its synthesis in the cytoplasm. In this study, to investigate the immediate intracellular responses following direct delivery of FUS protein, GFP-tagged FUS was delivered into the cytoplasm using SU10 for precise intracellular delivery of reagents, and live-cell imaging was performed with the CSU-W1 spinning disk confocal microscope.
As a result, we successfully visualized in real time the translocation of GFP-FUS from the cytoplasm to the nucleus and its subsequent formation of intranuclear structures. This result highlights the potential of the SU10–CSU combination as a powerful platform for direct protein delivery and real-time analysis of phase-separating protein dynamics without genetic manipulation.
| Video | |
 |
 |
Courtesy of Dr. Hitoki Nanaura (Nara Medical University), Dr. Taro Mannen (Ritsumeikan University), and Dr. Tomohide Saio (Tokushima University)
Experimental conditions
| Cell | U2OS |
| Substance | GFP-tagged Fused in Sarcoma protein (GFP-FUS) |
| Objective lens | 60x |
| Exposure time | 200 ms |
User’s voice
The combination of SU10-mediated intracellular delivery and high-speed imaging with CSU enabled us to visualize the intracellular behavior of the phase-separating protein FUS. We aim to apply this approach to studies on the mechanisms of neurodegenerative diseases.
Tracking Nuclear Delivery and Phase Separation Disruption by Artificial Antibodies (Monobodies)
Monobodies are artificially engineered antibody-like proteins. In this study, we used a monobody designed to bind molecules involved in the formation of nuclear droplets and to inhibit their liquid–liquid phase separation. Using SU10 for precise intracellular delivery of reagents, the monobody was directly delivered into the nucleus to evaluate whether intracellular liquid–liquid phase separation could be inhibited.
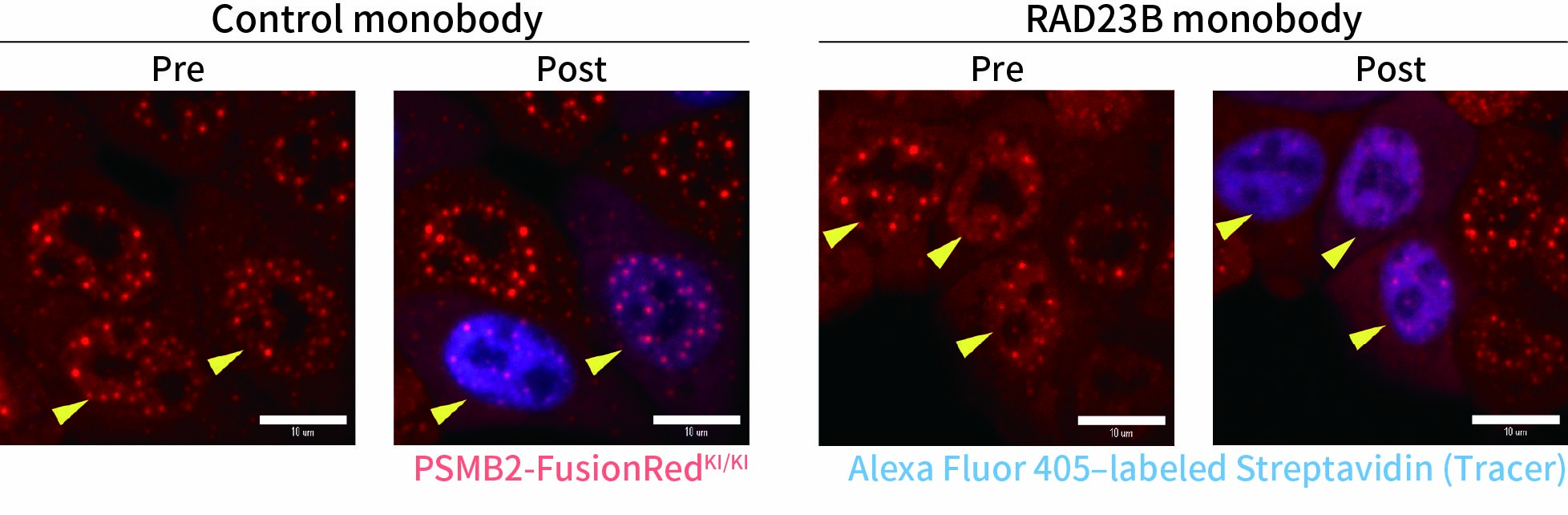
To capture immediate cellular responses following delivery, live-cell imaging was performed using the CSU-W1 spinning disk confocal microscope. In addition, a fluorescently labeled protein (Alexa Fluor™ 405–conjugated streptavidin) was co-delivered as a tracer to confirm successful solution delivery.
As a result, in cells delivered with a control monobody, nuclear droplets were still present after delivery (left image: Control monobody). In contrast, delivery of a monobody targeting the RAD23B led to the disappearance of droplet-like fluorescent signals (right image: RAD23B monobody and video).These results demonstrate that direct nuclear delivery of monobodies using SU10 enables direct inhibition of intracellular liquid–liquid phase separation.
| Video | |
 |
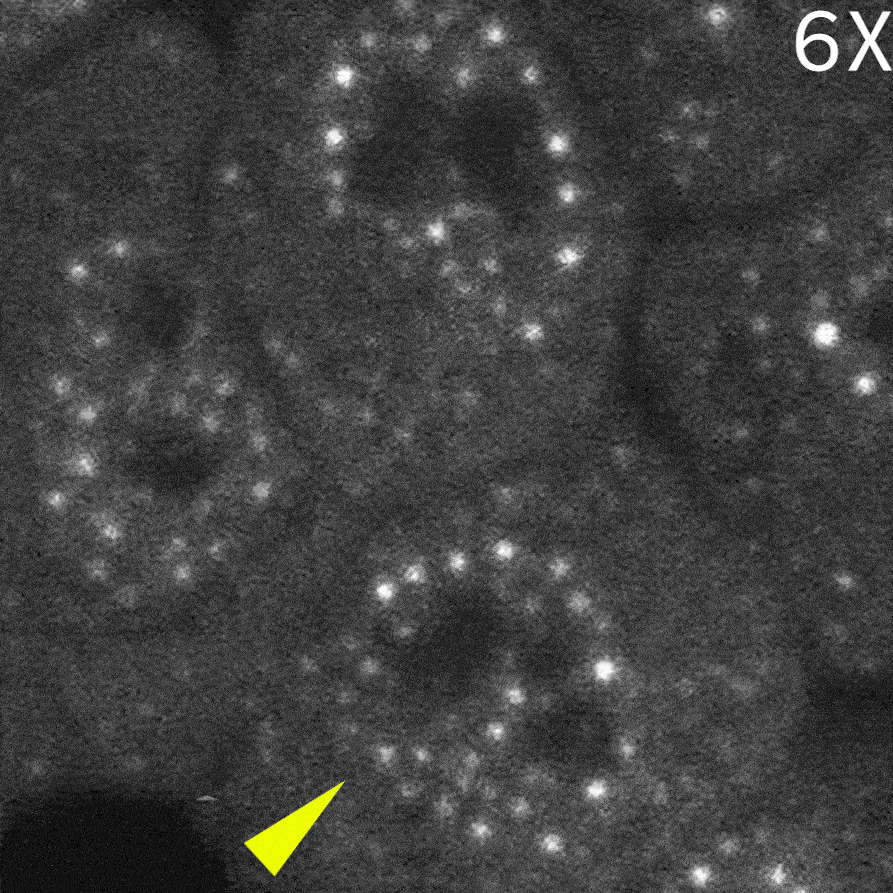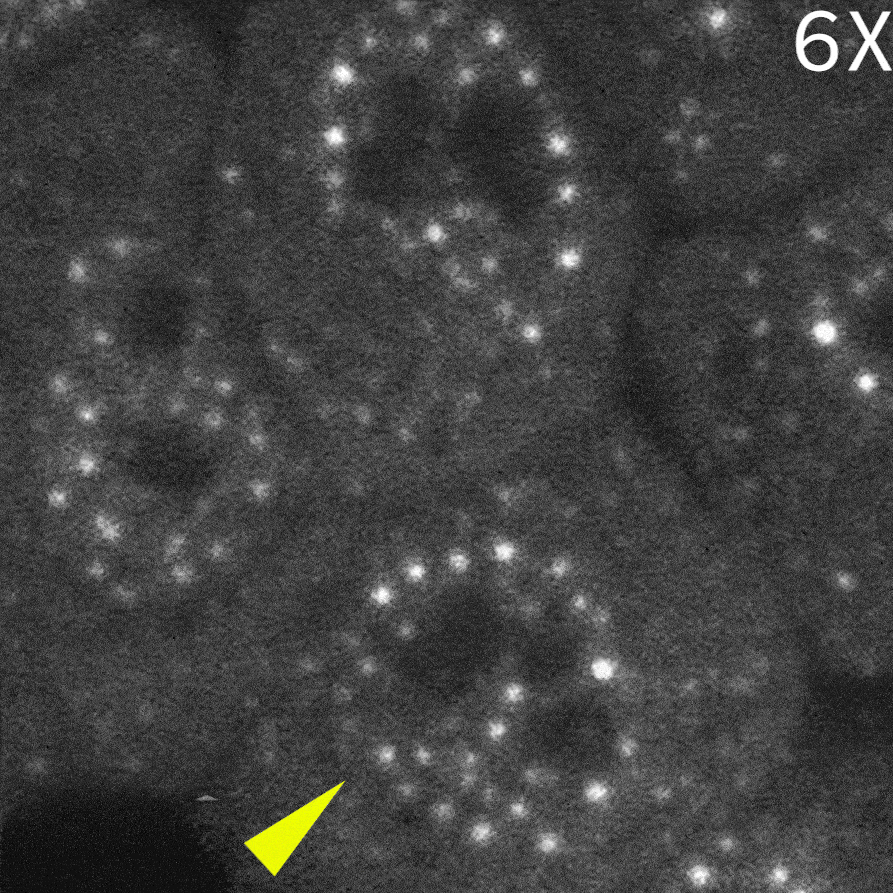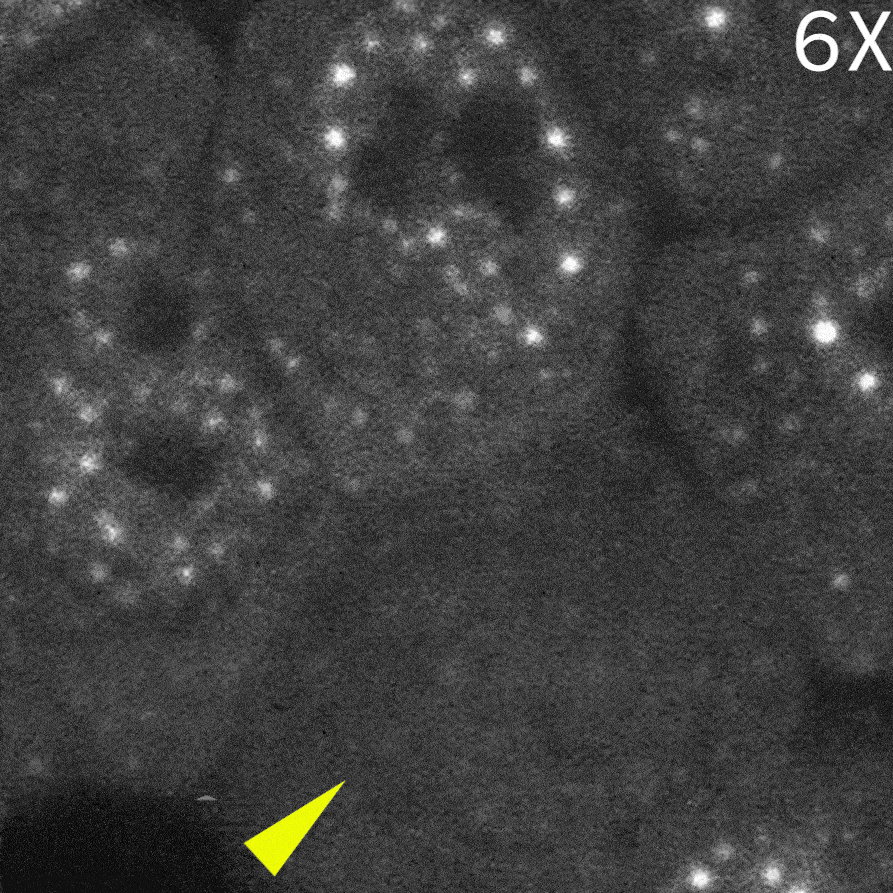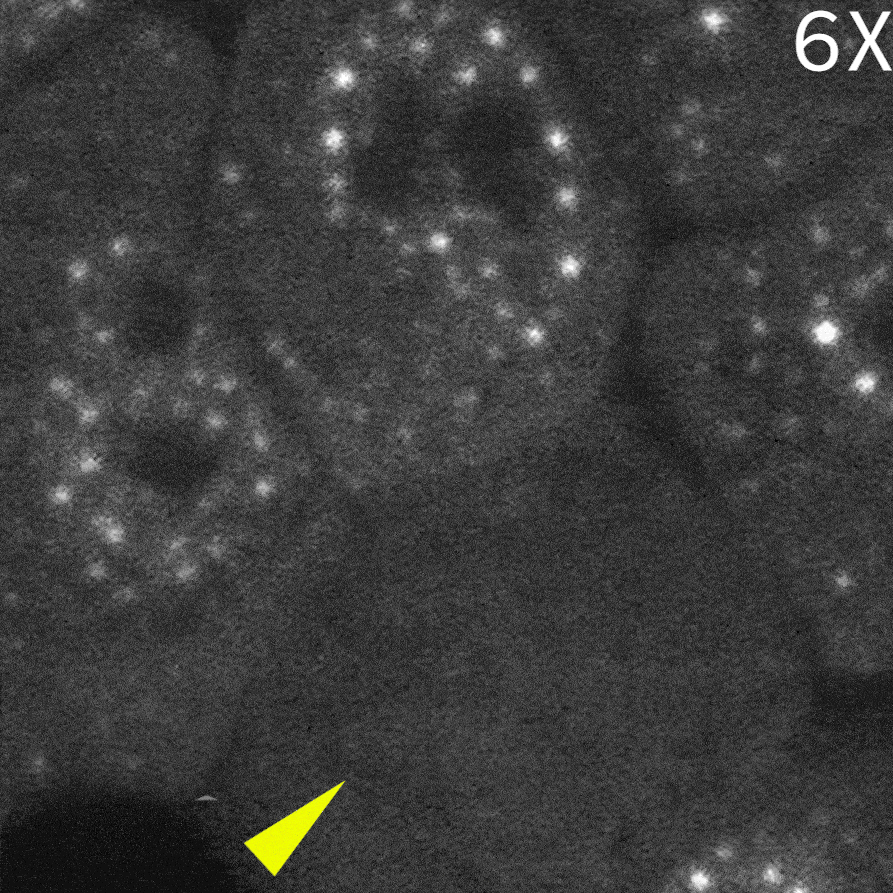 |
Courtesy of Dr. Yasushi Saeki (The Institute of Medical Science, The University of Tokyo) and Dr. Akinori Endo (Tokyo Metropolitan Institute of Medical Science)
Experimental conditions
| Cell | HCT-116 mutant strain |
| Substance | Monobodies & Alexa Fluor™ 405-conjugated streptavidin |
| Objective lens | 60x |
| Exposure time | 300 ms |
User’s voice
Small molecule artificial antibody monobodies are useful research tools for a variety of applications. However, their application to cell biological studies has been slow due to the difficulty of introducing them into cells. Using SU10, we were able to perform high-temporal resolution analysis by delivering monobodies directly into the cytoplasm and nucleus. We expect that SU10 will facilitate the application of useful research tools such as monobodies.
Tracking Antibody Delivery and Intracellular Molecular/Organelle Staining in Live Cells
SU10 enables the direct delivery of antibodies commonly used for immunostaining into living cells. In this experiment, a fluorescently labeled primary antibody against TOMM20, a protein localized to the mitochondrial outer membrane, was delivered into the cytoplasm using SU10 for precise intracellular delivery of reagents, followed by live-cell imaging with the CSU-W1 spinning disk confocal microscope.
Immediately after delivery, a strong fluorescent signal was observed around the delivery site. The signal then gradually diffused throughout the cytoplasm, and mitochondrial staining became clearly visible within a few minutes. Notably, this method does not require any membrane permeabilization or washing steps to remove excess antibodies, which are typically necessary in conventional immunostaining protocols.
These results demonstrate that the combination of direct antibody delivery using SU10 and high-speed live-cell imaging with CSU enables rapid and simple visualization of intracellular structures in living cells.
| Video | |
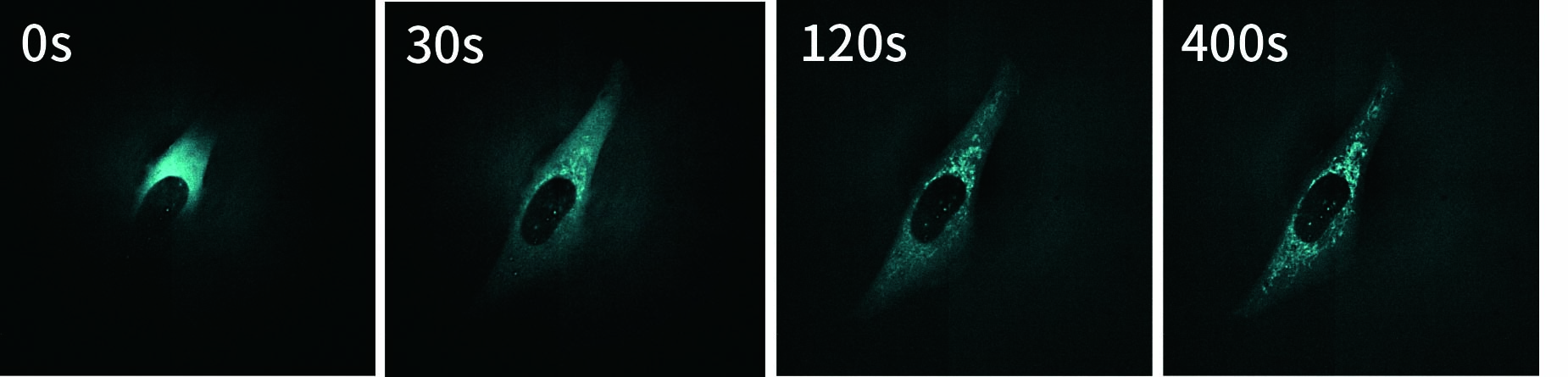 |
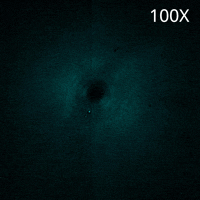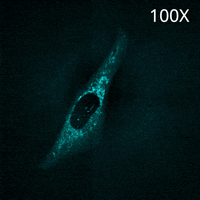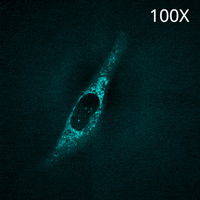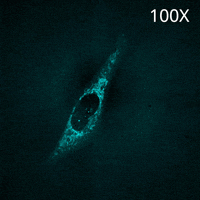 |
Experimental conditions
| Cell | HeLa |
| Substance | Alexa Fluor™ 647-conjugated anti-TOMM20 antibody, 0.25 mg/mL |
| Objective lens | 60x |
| Exposure time | 300 ms |
Tracking Compound Delivery and Nuclear Translocation
Fluorescent probes are valuable tools for real-time analysis of biological processes occurring inside cells; however, not all probes can permeate the membranes of living cells. SU10 enables the direct delivery of substances that are otherwise membrane-impermeable into the cytoplasm or nucleus of living cells. Therefore, by delivering fluorescent probes into cells using SU10, live-cell analysis can be performed easily without relying on membrane permeability.
In this experiment, to investigate the intracellular behavior of a compound that has histone-binding capability but low membrane permeability, the FITC-labeled compound was delivered into the cytoplasm using SU10 for precise intracellular delivery of reagents, followed by live-cell imaging with the CSU-W1 spinning disk confocal microscope.
As a result, immediately after cytoplasmic delivery, the compound rapidly translocated into the nucleus, where its binding to histones was visualized in real time. These results demonstrate that the combined use of SU10 and CSU enables real-time visualization of intracellular events occurring within just a few seconds.
| Video | |
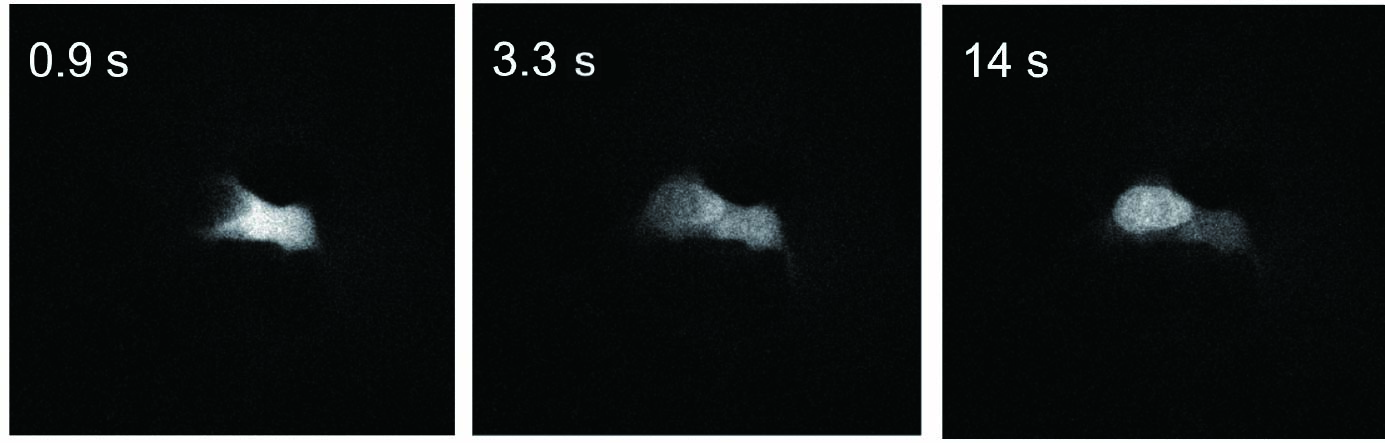 |
 |
Courtesy of Dr. Shigehiro Kawashima (Graduate School of Pharmaceutical Sciences, The University of Tokyo)
Experimental conditions
| Cell | HeLa S3 |
| Substance | Compound (FITC-labeled, low membrane permeability, histone-binding) |
| Objective lens | 20x |
| Exposure time | 50 ms at 180 ms intervals |
Related Products & Solutions
-
Nano-point Delivery/Nano-point Sampling Single Cellome™ Unit SU10
Single Cellome Unit SU10 enables the delivery of substances, such as recombinant proteins and genome editing tools, directly to the cytoplasm or nucleus of targeted single cells.
-
Spinning Disk Confocal CSU
Using our proprietary dual spinning disk design, Yokogawa’s confocal scanner units transform optical microscopes by enabling real-time live cell imaging.