Achieve Pharma Operational Excellence Through Data Integrity
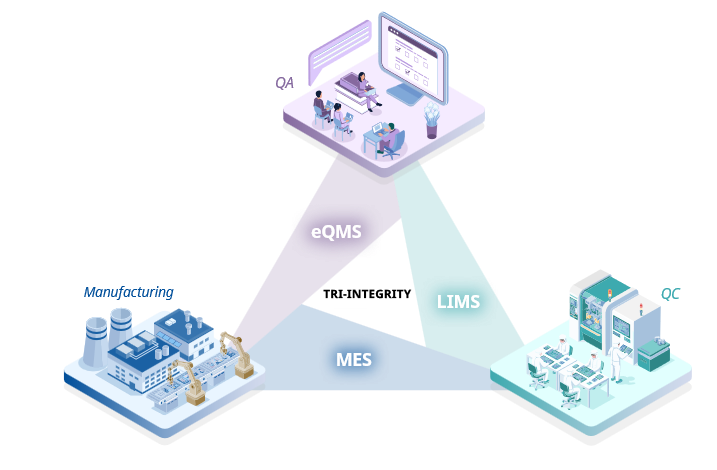
Discover how leading pharmaceutical manufacturers are transforming data into trusted, audit-ready insights. Connecting the three modular elements (MES, LIMS, eQMS) establishes the ultimate data integrity and traceability quality framework that extends beyond individual systems. This enables organizations to transform into continuously improving operations, creating a foundation for long-term success.
The Challenge Facing Pharma Today

Pharmaceutical manufacturers are under increasing pressure to ensure compliance, improve efficiency, and accelerate time to market—yet many are held back by disconnected systems and unreliable data.
- Siloed systems create multiple versions of the truth
- Manual processes increase compliance risk and audit exposure
- Limited traceability slows investigations and batch release
- Lack of real-time visibility delays critical decision-making
Without a strong data integrity foundation, even the best processes fall short
Connect. Trace. Transform. The Power of MES, LIMS, eQMS
Why Data Integrity Matters?
 Data integrity is more than a regulatory requirement—it’s the backbone of operational excellence. When your data is accurate, complete, and secure across its entire lifecycle, you can:
Data integrity is more than a regulatory requirement—it’s the backbone of operational excellence. When your data is accurate, complete, and secure across its entire lifecycle, you can:
Key Benefits
- Ensure consistent compliance with GMP and global regulations
- Maintain complete traceability for faster audits and investigations
- Reduce deviations and human error
- Enable faster, more confident decision-making
Click here to learn the 3 Pillars of Data Integrity and how achieving operational excellence starts with a structured approach.
Triple Pillars of Data Integrity for Pharma Operational Excellence


Upcoming Events
-
Event | Tradeshow Apr 14 - 15, 2026 San Francisco, CA2026 Generis American Biomanufacturing Summit (ABMS)
Join Yokogawa at the American Biomanufacturing Summit, where senior leaders across biomanufacturing, manufacturing technology, quality, compliance, and supply chain come together to share the trends and strategies shaping the industry’s future.
-
Event | Conference Jun 22 - 25, 2026 San Diego, CABio International Convention 2026
Join us at the BIO International Convention to explore how our Bioprocessing team is driving innovation in Bio Industrial Autonomy.
Details
Benefits
 Connecting the three modular elements (MES, LIMS, eQMS) establishes the ultimate data integrity and traceability quality framework that extends beyond individual systems. This enables organizations to transform into continuously improving operations, creating a foundation for long-term success.
Connecting the three modular elements (MES, LIMS, eQMS) establishes the ultimate data integrity and traceability quality framework that extends beyond individual systems. This enables organizations to transform into continuously improving operations, creating a foundation for long-term success.
Key Benefits
- Compliance driven by Data Integrity & Traceability
- Faster Batch Release
- Improved Productivity & Efficiency
- Robust and Efficient Change Control
- Pharma 4.0 Ready.
Better Decision Excellence begins by breaking down data silos between production, quality assurance, and quality control. Through integrated and shared information, organizations can enable consistent, data-driven GMP decision-making across the entire manufacturing lifecycle.
These improved decisions naturally lead to Batch Excellence. By eliminating data integrity risks caused by manual recording and fragmented documentation, manufacturers can accelerate batch production and validation while reducing errors and rework.
Building on this foundation, organizations can achieve Transformation Excellence. By embedding DX initiatives into on-site operations with appropriate validation and change control, sustainable digital transformation becomes possible—without compromising robust GMP compliance.
-
OpreX Quality Management System
Manage, monitor, and control quality assurance processes.
-
OpreX Laboratory Information Management System
OpreX Laboratory Information Management System(hereinafter "OpreX LIMS") is a Laboratory Information Management System (LIMS) package, which supports quality management operations. Furthermore, it standardizes quality management operations to reduce costs and improve the service level.
-
Manufacturing Operations Management for Pharmaceuticals
In pharmaceutical production, while the needs for pharmaceutical products are rapidly increasing, and manufacturing management is required to comply with laws and regulations, there is an urgent need for even higher quality and higher efficiency in order to survive in a severely competitive environment. Yokogawa is maximizing synergy between measurement, control, and information to meet the need for solutions to these issues.
Resources
BioPilot's flexibility empowers customized solutions for unique bioprocess needs, integrating diverse systems to improve speed, quality, reproducibility, and cost-effectiveness, ultimately preventing batch failures and supporting scalable innovation.
Downloads
Brochures
Videos
Discover how Yokogawa’s BioPilot transforms bioprocessing with seamless digital integration and intelligent automation. By unifying real-time data acquisition and advanced process control, BioPilot empowers scientists to streamline cell culture and purification workflows—enhancing reproducibility, accelerating development timelines, and enabling effective decisions in cell line, media, and process development.
More info: https://www.yokogawa.com/us/solutions/solutions/asset-operations-and-optimization/biopilot/
See how modern, cloud-based QMS delivers better compliance, visibility, and collaboration—watch now!
News
-
Press Release | Solutions & Products Feb 3, 2025 Yokogawa Releases OpreX Quality Management System to Accelerate DX in Quality Assurance Processes for the Manufacturing of Pharmaceuticals and Food & Beverages
- A cloud-based system that enables swift and flexible process improvements -
Looking for more information on our people, technology and solutions?
Contact Us